This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
Introduction
|
Polycystic ovary syndrome, commonly known as PCOS, is one of the most common hormonal disorders affecting women of reproductive age and it can also cause metabolic and overall health problems. Some symptoms that may be present with PCOS include infertility, obesity, and excess body hair but there are several other symptoms that may occur. [1] It’s likely that several biological systems are impacted in PCOS which is why treatment is limited to counteracting individual symptoms. To be diagnosed with PCOS, 2 out of the 3 following symptoms must be present: polycystic ovaries, irregular menstrual cycle, or excess testosterone. [8] As you may have noticed, it is possible to be diagnosed with polycystic ovary syndrome without having polycystic ovaries, which is why changing the disease’s name has been discussed in recent years. [3] My project is focused on the excess testosterone phenotype, as this is the underlying cause of some of the most disruptive and undesirable symptoms.
|
PCOS and StAR
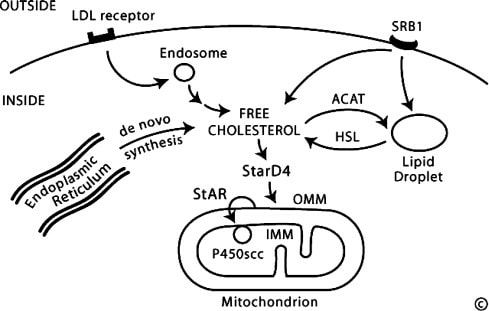
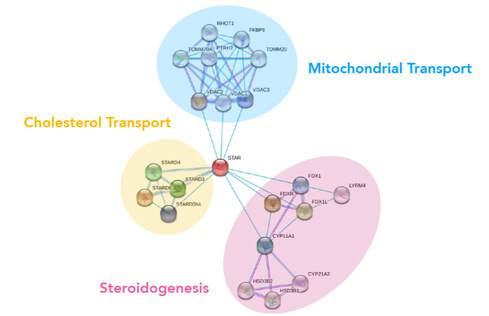
The Steroidogenic acute regulatory protein (StAR) has been implicated as a cause of PCOS because it is necessary for cellular synthesis of steroids. [2,3] StAR initiates the steroidogenesis process by transporting cholesterol - the precursor for steroids - within the mitochondrial membrane of cells. [2] Studies have shown that StAR is overexpressed in the ovarian cells of women with PCOS and rodent models of PCOS, yet what specific factors regulate this expression are still unknown. [3,4] StAR interacts with several other proteins that can be divided into 3 categories: mitochondrial transport, cholesterol transport, or steroidogenesis. These are all very much in-line with what is known about StAR function and its gene ontology.
Knowledge gap
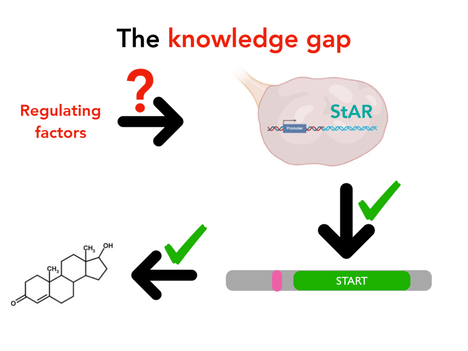
Currently, it is known that StAR is overexpressed in the ovaries of people and rats with PCOS [3,4] and we know that the protein transports cholesterol within the mitochondria to initiate testosterone biosynthesis. What is unknown is what factors regulate the expression of StAR. Previous, inconclusive studies looked at transcription factors that bind to the StAR promoter to impact expression.
|
Goals and hypothesis
The primary goal of my project is to better understand what factors regulate StAR expression and testosterone levels in PCOS, specifically at the level of transcription. I hypothesize that transcription factors will directly interact with the StAR promoter resulting in activation of the gene.
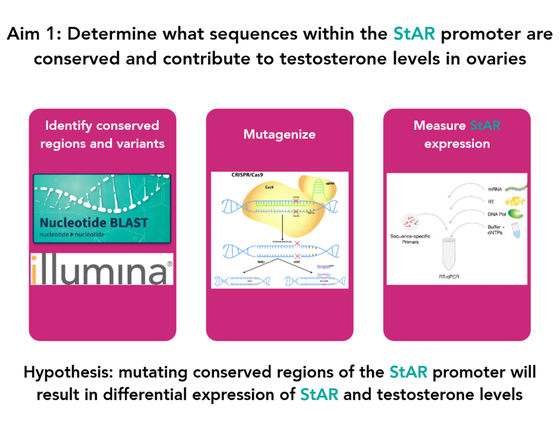
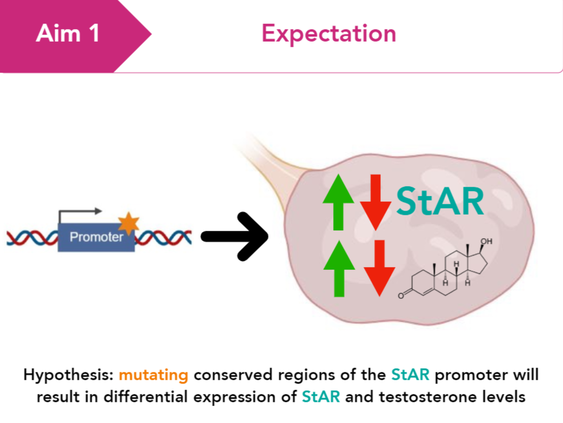
Aim 1: Determine what sequences within the StAR promoter are conserved and contribute to testosterone levels in ovaries
BLAST searches of StAR homologs and next-generation sequencing of rat lines will be performed to find StAR promoter sequence variants of conserved regions that correlate with differential testosterone levels in the ovaries of rats. CRISPR/Cas9 will be used to introduce sequence mutations of conserved regions into rat ovarian cell lines to observe effects of sequence variants on StAR expression using RT-PCR.
Evaluating phenotypes associated with variation within the StAR promoter allows for the confirmation that alterations of the StAR promoter impact regulation of StAR expression and testosterone levels.
I hypothesize that mutating conserved regions of the StAR promoter will result in differential expression of StAR and testosterone levels.
Evaluating phenotypes associated with variation within the StAR promoter allows for the confirmation that alterations of the StAR promoter impact regulation of StAR expression and testosterone levels.
I hypothesize that mutating conserved regions of the StAR promoter will result in differential expression of StAR and testosterone levels.
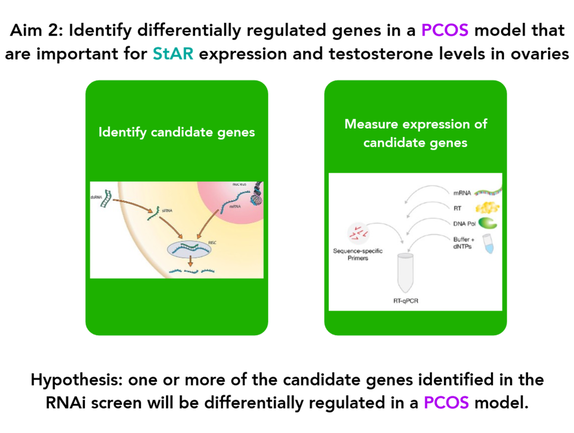
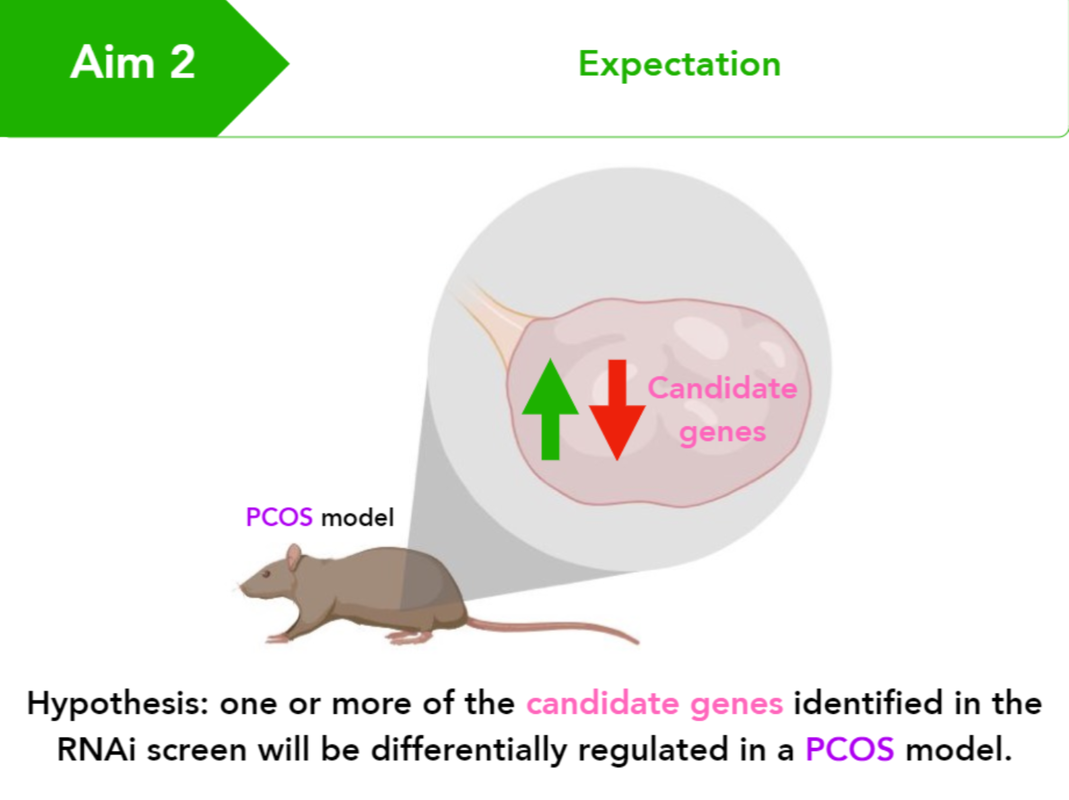
Aim 2: Identify differentially regulated genes in StAR mutants that are important for testosterone levels in ovaries
A genome-wide RNAi screen will be performed in rat ovarian cell lines and compared to controls to identify genes that impact StAR expression and testosterone levels in the ovaries. RT-PCR will be used to determine if prenatally androgenized rats display differential expression of any of the identified candidate genes.
Identifying which genes impact StAR expression and testosterone levels after knock-down with RNAi allows for confirmation that these genes are involved with regulating StAR. Identifying differentially regulated candidate genes in prenatally androgenized rats connects them with the PCOS phenotype specifically.
I hypothesize that one or more of the candidate genes identified in the RNAi screen will be differentially regulated in prenatally androgenized rats.
Identifying which genes impact StAR expression and testosterone levels after knock-down with RNAi allows for confirmation that these genes are involved with regulating StAR. Identifying differentially regulated candidate genes in prenatally androgenized rats connects them with the PCOS phenotype specifically.
I hypothesize that one or more of the candidate genes identified in the RNAi screen will be differentially regulated in prenatally androgenized rats.
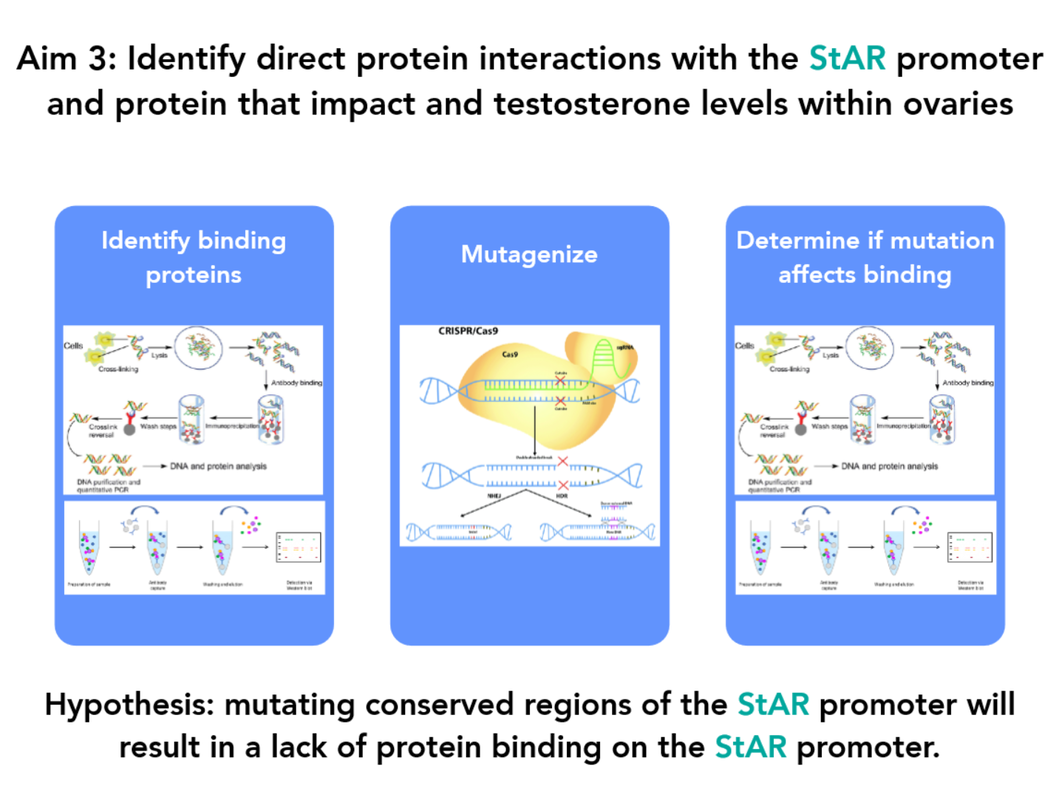
Aim 3: Identify direct protein interactions with the StAR promoter and protein that impact testosterone levels within ovaries
Chromatin immunoprecipitation (ChIP) and Co-immunoprecipitation (Co-IP) will be used to identify proteins that bind the StAR promoter or protein directly. Potential binding proteins will be based on candidate genes identified in Aim 2. CRISPR/Cas9 will then be used to mutate previously determined conserved regions of the StAR promoter and ChIP and Co-IP will be performed once again to determine if the previously identified proteins still bind the StAR promoter or protein itself.
Identifying proteins that bind the StAR promoter and have already been determined to be differentially expressed in StAR mutants will support their role in regulating StAR at the level of transcription as transcription factors. Identifying proteins that bind StAR directly will serve as a good foundation for future studies into other regulatory factors of StAR at the post-translational level.
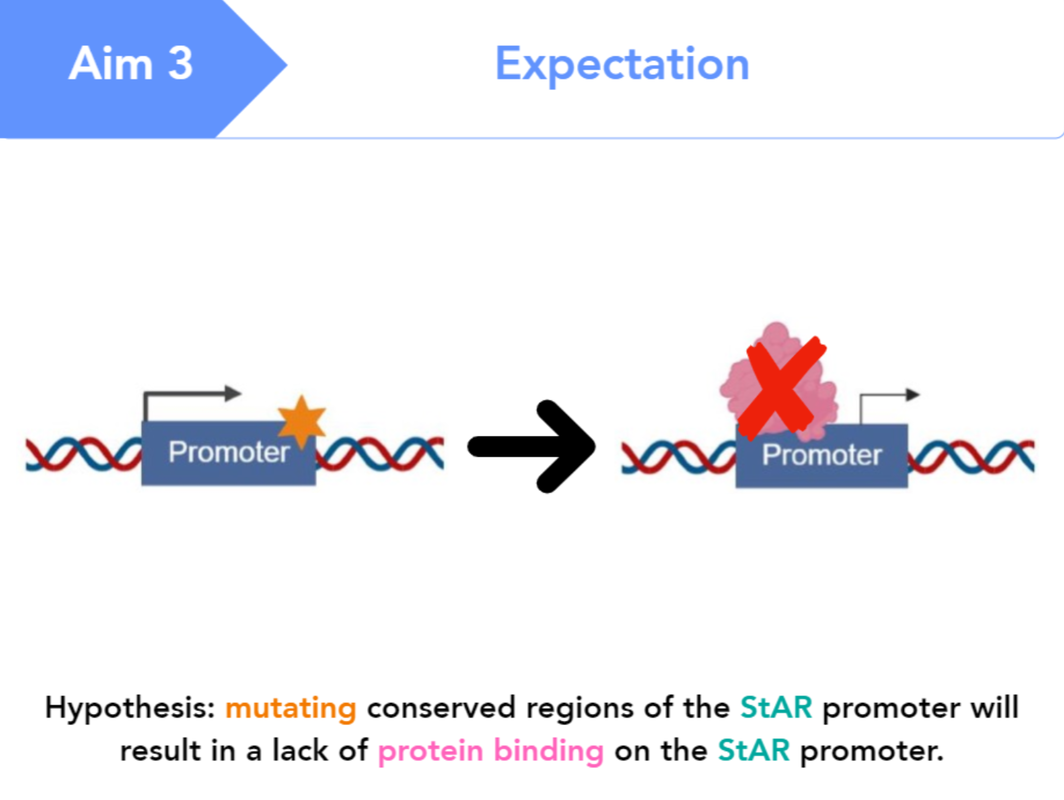
I hypothesize that mutating conserved regions of the StAR promoter will result in a lack of protein binding on the StAR promoter.
Identifying proteins that bind the StAR promoter and have already been determined to be differentially expressed in StAR mutants will support their role in regulating StAR at the level of transcription as transcription factors. Identifying proteins that bind StAR directly will serve as a good foundation for future studies into other regulatory factors of StAR at the post-translational level.
I hypothesize that mutating conserved regions of the StAR promoter will result in a lack of protein binding on the StAR promoter.
Conclusions and future directions
In summary, aim 1 will allow for confirmation that altering the StAR promoter impacts regulation of StAR expression and testosterone levels, aim 2 will identify candidate genes involved in regulating StAR, and aim 3 will identify proteins that directly interact with the StAR promoter and protein to regulate StAR expression. Investigating factors that regulate StAR expression and testosterone levels will guide the way in discovery of potential therapeutic targets. Since PCOS is such a common hormonal disorder with no current treatment plans that solve the underlying disease issues, discovery of new and effective therapies can benefit a lot of people. Future areas of research include looking into StAR regulating factors at the post-translational level and exploring the many other symptoms besides excess testosterone that are commonly experienced in PCOS.
Final and draft presentations
| sanderson_presentation_final_4.25.19.pdf | |
| File Size: | 5199 kb |
| File Type: | |
| sanderson_presentation_draft2_4.11.19.pdf | |
| File Size: | 3378 kb |
| File Type: | |
| sanderson_presentation_draft1_4.5.19.pdf | |
| File Size: | 2098 kb |
| File Type: | |
References
[1] Bednarska, S. & Siejka, A. (2017). The pathogenesis and treatment of polycystic ovary syndrome: What’s new? Advances in Clinical and Experimental Medicine. 26(2):359–367.
[2] Panda, P. K. et al. (2016). Genetics of PCOS: A systematic bioinformatics approach to unveil the proteins responsible for PCOS. Genome Data. 8:52-60.
[3] Kahsar-Miller, M. D. et al. (2001). Steroidogenic acute regulatory protein (StAR) in the ovaries of healthy women and those with polycystic ovary syndrome. American Journal of Obstetrics and Gynecology. 185(6):1381-1387
[4] Jahromi, M. S. et al. (2016). Elevated expression of steroidogenesis pathway genes; CYP17, GATA6 and StAR in prenatally androgenized rats. Gene. 593(1):167-171.
[5] Gillio-Meina, C. et al. (2003). GATA-4 and GATA-6 Transcription Factors: Expression, Immunohistochemical Localization, and Possible Function in the Porcine Ovary. Biology of Reproduction. 68(2):412-422.
[6] Jahromi, M. S. et al. (2018). Hypomethylation of specific CpG sites in the promoter region of steroidogeneic genes (GATA6 and StAR) in prenatally androgenized rats. Life Sciences. 207:105-109.
[7] Spinasanta, Susan. “It's Time to Rename Polycystic Ovary Syndrome.” Endocrine Web, 2016, www.endocrineweb.com/professional/meetings/its-time-rename-polycystic-ovary-syndrome.
[8] Trivax, B., & Azziz, R. (2007). Diagnosis of polycystic ovary syndrome. Clinical Obstetrics and Gynecology, 50(1):168–177.
[1] Bednarska, S. & Siejka, A. (2017). The pathogenesis and treatment of polycystic ovary syndrome: What’s new? Advances in Clinical and Experimental Medicine. 26(2):359–367.
[2] Panda, P. K. et al. (2016). Genetics of PCOS: A systematic bioinformatics approach to unveil the proteins responsible for PCOS. Genome Data. 8:52-60.
[3] Kahsar-Miller, M. D. et al. (2001). Steroidogenic acute regulatory protein (StAR) in the ovaries of healthy women and those with polycystic ovary syndrome. American Journal of Obstetrics and Gynecology. 185(6):1381-1387
[4] Jahromi, M. S. et al. (2016). Elevated expression of steroidogenesis pathway genes; CYP17, GATA6 and StAR in prenatally androgenized rats. Gene. 593(1):167-171.
[5] Gillio-Meina, C. et al. (2003). GATA-4 and GATA-6 Transcription Factors: Expression, Immunohistochemical Localization, and Possible Function in the Porcine Ovary. Biology of Reproduction. 68(2):412-422.
[6] Jahromi, M. S. et al. (2018). Hypomethylation of specific CpG sites in the promoter region of steroidogeneic genes (GATA6 and StAR) in prenatally androgenized rats. Life Sciences. 207:105-109.
[7] Spinasanta, Susan. “It's Time to Rename Polycystic Ovary Syndrome.” Endocrine Web, 2016, www.endocrineweb.com/professional/meetings/its-time-rename-polycystic-ovary-syndrome.
[8] Trivax, B., & Azziz, R. (2007). Diagnosis of polycystic ovary syndrome. Clinical Obstetrics and Gynecology, 50(1):168–177.